If Your Time Is Short
- A misleading graph posted on Facebook shows a spike in the number of reports made so far this year to the federal government’s Vaccine Adverse Event Reporting System, or VAERS, for children 6-17 years of age.
- Posters used the graph to suggest COVID-19 vaccinations are harming children. They also criticized California’s plans for a new vaccination mandate, which will require eligible schoolchildren to receive the shot once it gains full FDA approval.
- But health experts say VAERS data is voluntarily submitted and includes unverified information. An increase in reports doesn’t necessarily mean vaccinations are to blame for adverse events, especially since vaccinations are subject to rigorous testing before being rolled out to additional age groups.
- The Centers for Disease Control and Prevention has previously cautioned that VAERS results are not enough to determine whether a vaccine is harmful.
Moments after Gov. Gavin Newsom announced plans last month to mandate COVID-19 vaccination for all California schoolchildren, a graph often used to share misleading narratives on vaccinations started spreading across social media.
“This is the chart [for] 6-17 year olds for ALL VACCINES for all reactions over 30 years ... and @GavinNewsom says the covid vaccine is the same as all the others,” the tweet in early October, from a poster who does not appear to live in California, read.
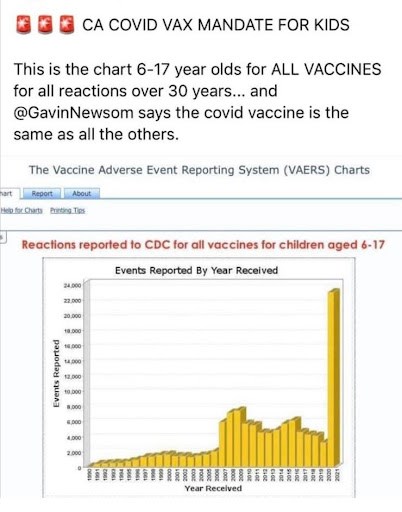
The tweet was shared more than 250 times. Screenshots quickly made their way to Facebook, where one poster urged parents to “follow the science” and “ask the questions.”
“They don't want people to know they are using our kids as science experiments,” one commenter wrote on the post.
Reacting to the post on Twitter, one commenter falsely described the vaccine as “a bioweapon.” In fact, the COVID-19 vaccines have undergone rigorous safety testing and federal health officials say they are safe and effective.
The Centers for Disease Control and Prevention graph includes unverified reports submitted by the general public to the federal government’s Vaccine Adverse Event Reporting System database, dating back to 1990 for children ages six to 17. The chart spikes in 2021, with more than 22,000 reports logged at the time screenshots of the CDC graph were shared on social media in early October.
It’s a real graph produced by the CDC. But does it prove COVID-19 vaccinations are unsafe for children, as the social media posts suggest?
With the CDC recommending this week that all children ages 5 through 11 get a low-dose COVID-19 vaccine, PolitiFact California decided to find out.
What Is VAERS?
The VAERS database was established in 1990 and is run by the Centers for Disease Control and Prevention, the agency that conducts public health research and issues guidance, and the Food and Drug Administration, which is tasked with monitoring new medications.
Researchers use it to collect data and assess vaccine safety. They analyze database reports to identify after-effects in new vaccines, find patient risk factors and monitor existing vaccinations. When dealing with severe adverse events, staff members may follow-up with reporting parties and ask for the patient’s medical records to learn more about what happened.
Healthcare providers and vaccine manufacturers are mandated to report adverse events to the database. But VAERS is open to everyone, so many reports also come from the public. The CDC encourages people to report adverse events after vaccination to the database, “even if it is not clear that the vaccine caused the adverse event.”
Because of this, the database website cautions that VAERS data should be interpreted with these limitations in mind.
“The number of reports alone cannot be interpreted or used to reach conclusions about the existence, severity, frequency, or rates of problems associated with vaccines,” the VAERS website states.
The Graph
There have been approximately 24,000 reports to the database so far this year. But those reports are not just for children who received a COVID-19 vaccination.
The graph shows reports tied to all childhood vaccines, so some are not related to COVID-19 vaccinations. Still, most of the reports — about 22,000 — cite a COVID-19 shot.
Experts say there are a variety of reasons why that number might be so high. Dean Blumberg, chief of pediatric infectious diseases at UC Davis Children’s Hospital in Sacramento, said as vaccine hesitancy has risen in recent years, reports to the database have gone up.
“When a new vaccine comes out and the anti-vaccine lobby starts to attack it, then there is a spike in VAERS reports,” Blumberg said. “We saw this most dramatically with HPV vaccine approval and all the pushback against the manufacturer trying to push through school mandates in many states. And it seems reasonable to expect the same with COVID vaccines, especially considering how everything related to COVID has become politicized.”
Blumberg also said publicity and media coverage drive reports to the database.
“There is more awareness to report any possible vaccine reactions,” he said. “Anyone can make a report, medical professionals or parents.”
David Lo, a professor of biomedical sciences and senior associate dean of research at UC Riverside’s School of Medicine, stressed that the large number of reports does not mean COVID-19 vaccines are harmful.
“Any drug or vaccine is continually monitored for safety,” Lo said. “The safety profile of vaccines continues to be very strong, and testing and monitoring are especially rigorous.”
Vaccine Testing
To be approved for children, COVID-19 vaccines have to go through a lengthy process. After initial research and development, vaccinations are tested in humans in three phases, each with an increasing number of people.
Currently, the Pfizer-BioNTech vaccine is the only COVID-19 vaccination approved for children under the age of 16. In October, the FDA granted it emergency use authorization. That means it followed an expedited process to approval because of the public health emergency the pandemic triggered. But it doesn’t mean the process skipped important steps or ignored safety measures. For emergency use authorization, vaccines must meet the FDA’s standards for safety, effectiveness and manufacturing quality.
Pfizer-BioNTech submitted a study of 2,264 subjects to the FDA before receiving approval for its vaccine to be used in 12 to 15-year-olds. About half of the study participants received the vaccine and half received a placebo.
Among study participants that received the vaccine, the immune response against COVID-19 was at least as high as that of young adults, and adverse effects were also comparable to older age groups. The most common symptoms were fatigue, fever, headache, chills and injection-site pain, which usually occurred after participants received the second dose of the vaccine.
Out of the roughly 1,100 people who received the vaccine in the study, five serious adverse events were reported, but all were determined to be unrelated to the vaccination.
“These serious adverse events encompassed medical events occurring at a frequency similar to that in the general population aged 12–15 years, with none considered to be related to vaccination,” the study reads. “No specific safety concerns were identified among adolescent vaccine recipients.”
Two months after the study, none of the vaccinated participants had gotten ill with COVID-19. Eighteen of the people in the placebo group had contracted the virus.
Pfizer recently completed a similar study of 5 to 11-year-old children, and received emergency use authorization in young children from the FDA earlier this week. That decision was affirmed by the CDC on Tuesday evening, clearing the way for children to begin receiving shots.
The study of more than 4,600 children worldwide showed more than 90% protection against symptomatic COVID-19 among young children, who will receive one-third the dose of adults. In the trial, young children were less likely to experience side effects like fever than older children, teenagers and adults. Lo said the “hard facts” of the Pfizer studies, particularly the results that show the vaccine’s effectiveness, should assure parents that the shot is safe for children and motivate them to get their kids vaccinated.
Concerns have been raised about the COVID-19 vaccine’s connection to young people developing myocarditis, an inflammation of the heart muscle.
NPR reported the CDC has confirmed 877 cases of myocarditis after administration of the Pfizer or Moderna vaccine in people age 30 or younger, but there have been no deaths, said Dr. Matthew Oster, who studies myocarditis for the CDC and is a pediatric cardiologist at Children's Healthcare of Atlanta. He explained that COVID-19 itself can cause MIS-C, a type of myocarditis, and other heart-related issues.
"The bottom line is getting COVID, I think, is much riskier to the heart than getting this vaccine," Oster said.
Our Ruling
The initial tweet and subsequent Facebook posts suggested that COVID-19 vaccines are not safe for children and displayed a graph of data from the federal Vaccine Adverse Event Reporting System.
The VAERS database is intended to collect data on adverse events after vaccination that may or may not be linked to the vaccine a person received. Experts analyze and verify those reports to identify trends in after-effects and patient risk factors, as well as monitor vaccines that have been available for decades. The database includes a disclaimer that says the number of reports about a specific vaccine should not be used to draw conclusions about vaccine safety.
Experts say that as the anti-vaccination movement has grown in recent years, the database has seen increasing numbers of misleading reports. More publicity of the database has also contributed to more people reporting adverse events.
Experts say COVID-19 vaccinations are tested rigorously and monitored as they are released to the public. Furthermore, scientific studies of thousands of children have determined that serious adverse events after vaccination are rare and that vaccinations are effective against COVID-19.
While the graph is a real representation of reports submitted to VAERS, the social media posts have greatly distorted its meaning and falsely suggested that the COVID-19 vaccines are harmful for children. Therefore, we rate the posts Mostly False.
MOSTLY FALSE – The statement contains an element of truth but ignores critical facts that would give a different impression.
Sources:
Facebook post, Oct. 10, 2021
Twitter post, Oct. 1, 2021
PolitiFact, Federal VAERS database is a critical tool for researchers, but a breeding ground for misinformation, May 3, 2021
Office of the Governor, California Becomes First State in Nation to Announce COVID-19 Vaccine Requirements for Schools, Oct. 1, 2021
CNN, 5- to 11-year-olds can now get a Covid-19 vaccine in the US, Nov. 2, 2021
Vaccine Adverse Event Reporting System, About VAERS
Vaccine Adverse Event Reporting System, VAERS Data
Vaccine Adverse Event Reporting System, Frequently Asked Questions (FAQs)
U.S. Food and Drug Administration, Emergency Use Authorization for Vaccines Explained
U.S. Food and Drug Administration, FDA Vaccine Facts: The Path for Vaccines from Research to FDA Approval
Morbidity and Mortality Weekly Report (U.S. Department of Health and Human Services/Centers for Disease Control and Prevention), The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine in Adolescents Aged 12–15 Years — United States, May 2021, May 21, 2021
Pfizer, Pfizer and BioNTech submit initial data to the U.S. FDA from pivotal trial of COVID-19 vaccine in children 5 to <12 years of age, Sept. 28, 2021
Email interview with Dean Blumberg, chief of pediatric infectious diseases at UC Davis Children’s Hospital, Oct. 19, 2021
Email interview with David Lo, professor of biomedical sciences and senior associate dean of research at UC Riverside’s School of Medicine, Oct. 18, 2021